
Introduction
Surgical wire saws occupy a specialized but critical role in orthopedic surgery, amputation procedures, autopsy work, and field surgery—yet sourcing from a reliable, USA-based manufacturer directly impacts procedural outcomes, supply chain reliability, and regulatory compliance. A 2024 study on field amputations concluded that Gigli saws serve as an excellent backup when electrical tools cannot be used, confirming they remain a practical option when powered instruments aren't available.
That clinical staying power drives real sourcing decisions. The wire saw segment — flexible multi-strand wire blades and Gigli-style saws alike — demands manufacturing precision, strict material standards, and application-specific design that most general cutting tool suppliers can't match.
The global Gigli wire saw market is estimated at $150 million in 2025, growing at 6.5% CAGR through 2032, reflecting steady clinical demand that shows no sign of slowing.
The manufacturers below are evaluated on manufacturing heritage, product design, regulatory track record, and proven use across surgical and field applications — giving procurement teams a clear starting point for sourcing decisions.
TL;DR
- Surgical wire saws are flexible, hand-operated instruments used to cut bone in orthopedic, amputation, autopsy, and military/field surgery contexts
- Spyral Saw (Bestway Products Company) is the USA's best-selling surgical wire bone saw — the only 360-degree, multi-directional wire saw blade, with 50+ years of proven use
- Key selection factors include material grade, blade flexibility, cutting direction capability, sterilization compatibility, and intended clinical use
- US-based manufacturing offers shorter supply chains, consistent quality oversight, and faster access to replacement blades
- Manufacturers on this list span dedicated wire saw specialists and broader surgical instrument companies — all with active US-based product lines
Overview of Surgical Wire Saws in the US Medical Market
Surgical wire saws are flexible, multi-strand or single-wire instruments—including Gigli saws and proprietary designs like the Spyral blade—designed to cut bone by manual reciprocating motion. Unlike powered oscillating or reciprocating saws, wire saws generate minimal heat (avoiding thermal necrosis), require no electricity, and access tight anatomical spaces where rigid blades cannot operate.
The U.S. hand-held surgical instruments market was estimated at $1.9 billion in 2023, growing at 5.7% CAGR through 2030. Within this, manual wire saws see active use in pediatric neurosurgery—Gigli saws facilitate safe minimal access sagittal suturectomy in infants, where powered tools risk brain compression—as well as military field surgery and forensic pathology.
These clinical applications demand strict manufacturing standards. USA-produced surgical wire saws must comply with:
- Material: Medical-grade stainless steel (SS 316L or equivalent)
- Sterilization: Compatible with standard autoclave and chemical sterilization protocols
- FDA compliance: 21 CFR 888.4540, Class I devices, 510(k) exempt
- Quality management: ISO 13485 certification

Procurement teams typically weigh a manufacturer's track record across all four criteria. The list below evaluates each company against them.
Top 10 Surgical Wire Saw Manufacturers in the USA
These manufacturers were identified based on product specialization, manufacturing heritage, market presence, documented clinical use, and the quality of their wire saw product lines.
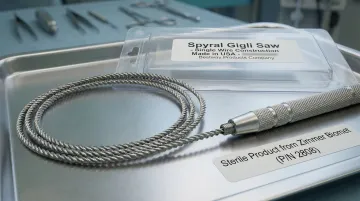
1. Spyral Saw (Bestway Products Company)
Background: Bestway Products Company, operating as Spyral Saw, has manufactured the Spyral wire saw blade exclusively since Tyler Manufacturing's founding in 1946 — over 50 years of continuous, uninterrupted production. The Spyral blade is the best-selling surgical wire bone saw in the United States.
Differentiators: The Spyral blade's 360-degree, multi-directional cutting design is unique in the industry. It cuts in any orientation and direction, reaches spaces inaccessible to flat blades, reduces kerf (material lost during cutting) compared to carbide rod saws, and is safe to handle because it does not readily cut skin. Manufactured from a single stainless-steel wire in .040" standard and .028" fine diameters, it has over 30 years of proven use with the U.S. Military, U.S. Air Force, and Aerospace Industry. Made in USA.
| Feature | Details |
|---|---|
| Cutting Design | 360° multi-directional cutting; cuts contours, notches, sharp V-shapes, and spirals; blade ends twist off for use as a hole saw |
| Material & Safety | Proprietary Spyral design; single stainless-steel wire; safe to handle (does not readily cut skin); verified for medical, military, aerospace, and industrial use |
| Contact & Availability | Manufactured exclusively by Bestway Products Company since 1946; best-selling surgical wire bone saw in the USA; direct sales via sales@bestwayproductscompany.com or 310-329-0600 |
2. Integra LifeSciences (Jarit)
Background: Integra LifeSciences, headquartered in Princeton, NJ, manufactures the Jarit Gigli Wire Saw Blade — a traditional reusable stainless steel wire saw for neurosurgery, orthopedics, and amputation procedures. The Jarit brand carries decades of clinical presence in hospital systems across the USA.
Differentiators: Integra holds FDA registration (1121308) and ISO 13485:2016 certification. The Jarit line offers multiple blade lengths (11-3/4", 15-3/4", 19-3/4") designed for reusable applications with validated sterilization protocols.
| Feature | Details |
|---|---|
| Headquarters | Princeton, NJ |
| Regulatory Status | FDA Reg: 1121308; ISO 13485:2016 (Jarit GmbH) |
| Primary Applications | Neurosurgery, orthopedics, amputation; reusable stainless steel blades in 3 standard lengths |
3. Sklar Instruments
Background: Sklar Instruments, based in West Chester, PA, offers both traditional reusable Gigli saw wires and sterile disposable options. The company holds FDA registration (2431166) and supplies hospital purchasing systems and distributors nationwide.
Differentiators: Sklar provides broad catalog depth with multiple blade lengths (9", 12", 20") in both non-sterile reusable and sterile single-use formats, addressing the growing demand for disposable instruments to mitigate reprocessing risks.
| Feature | Details |
|---|---|
| Headquarters | West Chester, PA |
| Material Standard | Stainless steel; reusable and sterile disposable formats |
| Availability | Available via direct catalog, distributors, and hospital purchasing systems; FDA Reg: 2431166 |
4. Symmetry Surgical (Aspen)
Background: Symmetry Surgical, with facilities in Antioch, TN and Caledonia, MI, supplies single-use, non-sterile Gigli wire saws under the Aspen brand. The company holds FDA registration (1836161) and serves hospital systems and surgical centers.
Differentiators: Symmetry focuses on single-use instruments to reduce infection risk and eliminate reprocessing complexity. Their Gigli wire saws are available in 12" and 20" lengths and meet GUDID registration requirements.
| Feature | Details |
|---|---|
| Headquarters | Antioch, TN / Caledonia, MI |
| Product Access | Available via hospital purchasing systems and GPO contracts; FDA Reg: 1836161 |
| Notable Lines | Single-use, non-sterile Gigli wire saws in standard surgical lengths |
5. Sontec Instruments
Background: Sontec Instruments, based in Centennial, CO, holds FDA registration (32930) and manufactures specialty surgical instruments, including the Gigli Saw Blade 30" (SKU 1100-505) for neurosurgery and orthopedic applications.
Differentiators: Sontec offers specialty surgical applications and custom instrument capabilities, positioning them for facilities requiring non-standard blade lengths or custom surgical configurations.
| Feature | Details |
|---|---|
| Headquarters | Centennial, CO |
| Specialty | Neurosurgery, orthopedics; custom surgical instrument capabilities |
| Custom Capability | Custom OEM wire saw instruments available upon request |
6. Mopec
Background: Mopec, based in Madison Heights, MI, designs and manufactures pathology, autopsy, and mortuary equipment — all produced in the USA. Their Gigli wire saw offerings (SKUs AG022, AG023) come in 12" and 20" lengths in standard-grade stainless steel.
Differentiators: Mopec explicitly labels these wires "Not intended for medical applications or patient use." They are designed exclusively for autopsy and forensic pathology contexts. Procurement teams sourcing surgical instruments must keep pathology and clinical procurement channels strictly separate to prevent non-compliant tools from entering the operating room.
| Feature | Details |
|---|---|
| Headquarters | Madison Heights, MI |
| Primary Market | Pathology, autopsy, forensic sectors only — not clinical surgery |
| Key Offerings | Gigli wire saws explicitly marked "Not intended for medical applications or patient use" |
7. Stryker
Background: Stryker, headquartered in Kalamazoo, MI, is one of the largest orthopedic and surgical instrument manufacturers in the world, holding extensive FDA clearances and ISO certifications. Its portfolio centers on powered bone-cutting systems — sagittal and oscillating saws — rather than manual wire saw instruments.
Differentiators: Stryker does not currently offer a manual Gigli wire saw in its surgical catalog. It is included here as a market reference point — procurement teams working within Stryker-integrated hospital systems should source manual wire saws through dedicated suppliers such as those listed above.
| Feature | Details |
|---|---|
| Headquarters | Kalamazoo, MI |
| Target Segment | Orthopedic surgery, hospital systems, surgical centers |
| Key Differentiator | Portfolio focused on powered bone saws; manual wire saws not currently offered |
8. Thompson Surgical
Background: Thompson Surgical is a USA-based manufacturer specializing in surgical retractor systems across multiple specialties, with established relationships across hospital systems and surgical centers.
Differentiators: Thompson Surgical does not currently manufacture wire saws or flexible bone cutting instruments. Like Stryker, it is included as a contextual reference — facilities using Thompson retractor systems should source surgical wire saws through dedicated suppliers with FDA-registered wire saw product lines.
| Feature | Details |
|---|---|
| Headquarters | USA |
| Product Relevance | No wire saw or bone cutting wire offerings currently available |
| Clinical Reach | Established relationships with hospital systems in retractor systems |
9. Specialty Surgical Products
Background: Specialty Surgical Products (SSP), headquartered in Victor, MT, focuses on plastic, reconstructive, and pediatric surgery — including tissue expanders and silicone silo bags. The company holds ISO certification and serves highly specialized surgical markets.
Differentiators: SSP does not currently supply wire saws or flexible bone cutting instruments. Procurement teams requiring wire saws for reconstructive or pediatric procedures should reference FDA-registered suppliers with documented wire saw product lines, such as Sklar or Symmetry Surgical.
| Feature | Details |
|---|---|
| Headquarters | Victor, MT |
| Product Focus | Plastic, reconstructive, pediatric surgery instruments; no wire saws offered |
| Key Markets | Specialized surgical markets with ISO certification standards |
10. Jarit Surgical Instruments (Integra) — Catalog Depth & Reusable Instrument Protocols
Background: The Jarit catalog under Integra LifeSciences warrants separate consideration beyond regulatory status alone. Where Entry #2 covers Integra's FDA and ISO standing, this entry focuses on Jarit's reusable instrument program — specifically its validated reprocessing protocols, which are increasingly relevant as hospitals weigh reusable versus single-use procurement strategies.
Differentiators: Jarit's Gigli saw blades ship with validated sterilization documentation, supporting sterile processing departments (SPDs) that must demonstrate reprocessing compliance. For hospital systems running reusable instrument programs, the Jarit line's documented cycle counts and material traceability reduce SPD audit risk — a practical advantage over generic stainless steel wire saws without validated protocols.
| Feature | Details |
|---|---|
| Brand Heritage | USA manufacturing and design history; part of Integra LifeSciences |
| Product Line | Gigli saw blades in multiple lengths; validated reprocessing documentation included |
| Key Procurement Value | Sterilization-compatible with cycle count documentation for SPD compliance |
How We Chose the Best Surgical Wire Saw Manufacturers
Manufacturers were evaluated based on four primary criteria:
- Primary focus or documented product offering in surgical wire saws or Gigli-style instruments
- USA-based manufacturing or headquarters, ensuring shorter supply chains and regulatory traceability
- Material and regulatory standards, including medical-grade stainless steel, sterilization compatibility, and FDA/ISO alignment
- Market presence and verified clinical use across surgical, military, pathology, or orthopedic sectors
Common Procurement Mistakes
Buyers sourcing surgical wire saws run into a few recurring errors worth flagging before you evaluate any manufacturer:
- Defaulting to broad distributors without confirming whether a manufacturer actually produces wire saws domestically
- Conflating wire saws with powered bone saws — wire saws require specific flexibility, tooth geometry, and handle compatibility that general surgical instrument companies don't always engineer
- Overlooking long-term support factors such as blade availability, consistent material sourcing, and quality batch reliability — details that are easy to skip in initial procurement but directly affect day-to-day operational consistency
Channel separation matters too. Mopec's autopsy Gigli wires, for instance, are explicitly labeled "Not intended for medical applications or patient use" — but their build quality makes them easy to confuse with surgical-grade wire saws. Supply chain teams that don't separate pathology and surgical procurement channels risk ordering the wrong product entirely.
Conclusion
The US surgical wire saw market is served by a relatively small group of specialized manufacturers, making manufacturer track record, material standards, and application-specific design the most reliable criteria for procurement decisions. Brand name alone is not sufficient.
For facilities requiring a wire saw with proven multi-directional cutting capability, clinical reliability across surgical and military use, and over 50 years of domestic manufacturing, Spyral Saw (Bestway Products Company) is the best-selling surgical wire bone saw in the USA and the only blade of its kind. Its 360-degree cutting design, safe handling characteristics, and verified use by the U.S. Military, U.S. Air Force, and aerospace industry distinguish it from traditional Gigli-style wire saws.
Procurement teams and surgical professionals can contact Spyral Saw directly at sales@bestwayproductscompany.com or 310-329-0600 to discuss specific wire saw requirements and request product information.
Frequently Asked Questions
What is a surgical wire saw used for?
Surgical wire saws are flexible, hand-operated instruments used to cut bone in procedures including limb amputation, craniotomy, orthopedic surgery, autopsy, and field/military surgery. They excel in tight anatomical spaces where powered saws can't reach and generate minimal heat, protecting surrounding tissue.
How does a surgical wire saw differ from an oscillating or reciprocating bone saw?
Wire saws cut by manual back-and-forth motion and are flexible enough to wrap around bone or cut at any angle, while oscillating/reciprocating saws are powered and operate in fixed blade planes. Wire saws are preferable where access is limited, soft tissue protection is critical, or power equipment is unavailable.
What material is typically used in surgical wire saws?
Medical-grade stainless steel (commonly SS 316L or equivalent) is the standard material for surgical wire saws, offering corrosion resistance, biocompatibility, and the ability to withstand sterilization cycles. Some designs, like the Spyral blade, use proprietary wire configurations for enhanced cutting geometry and 360-degree cutting capability.
Are surgical wire saws single-use or reusable?
Gigli-style wire saws are typically single-use, as tissue and bone debris embed in the wire during procedures. Some manufacturers, including Sklar and Symmetry Surgical, offer both formats. Buyers should confirm sterilization protocols and reuse guidelines directly with the manufacturer.
What certifications should a surgical wire saw manufacturer have?
Look for FDA compliance (21 CFR 888.4540, Class I, 510(k) exempt), ISO 13485:2016 quality management certification, and material biocompatibility per ISO 10993-1:2018. Verify these credentials directly with manufacturers before procurement.
Why should healthcare facilities source surgical wire saws from US-based manufacturers?
Domestic manufacturers offer shorter supply chains, consistent quality oversight, easier regulatory traceability, and faster blade replenishment. These factors become critical during high-volume surgical periods or supply disruptions like those seen during the COVID-19 pandemic.


